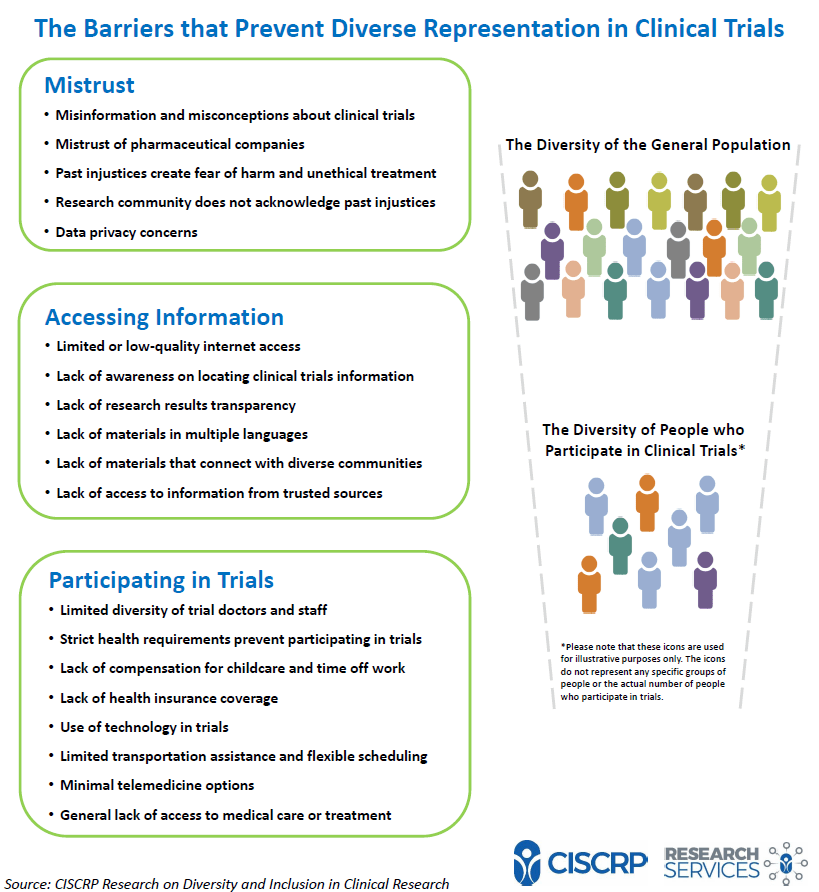
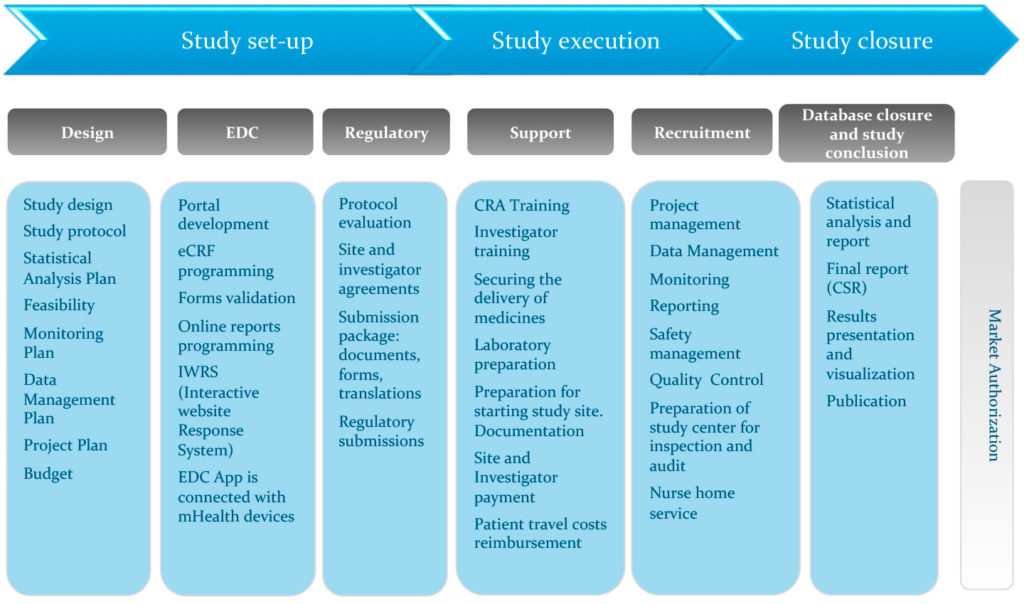
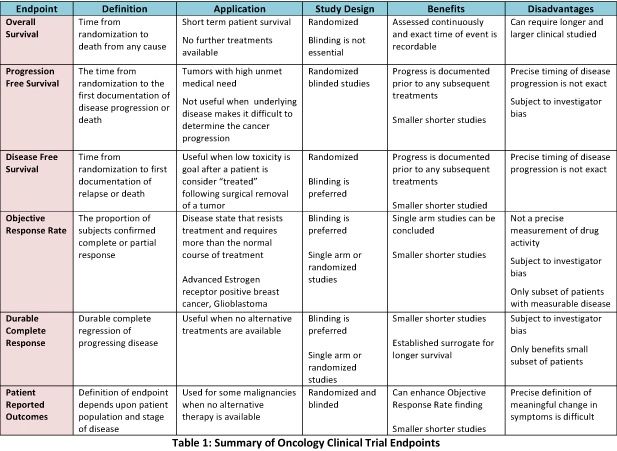
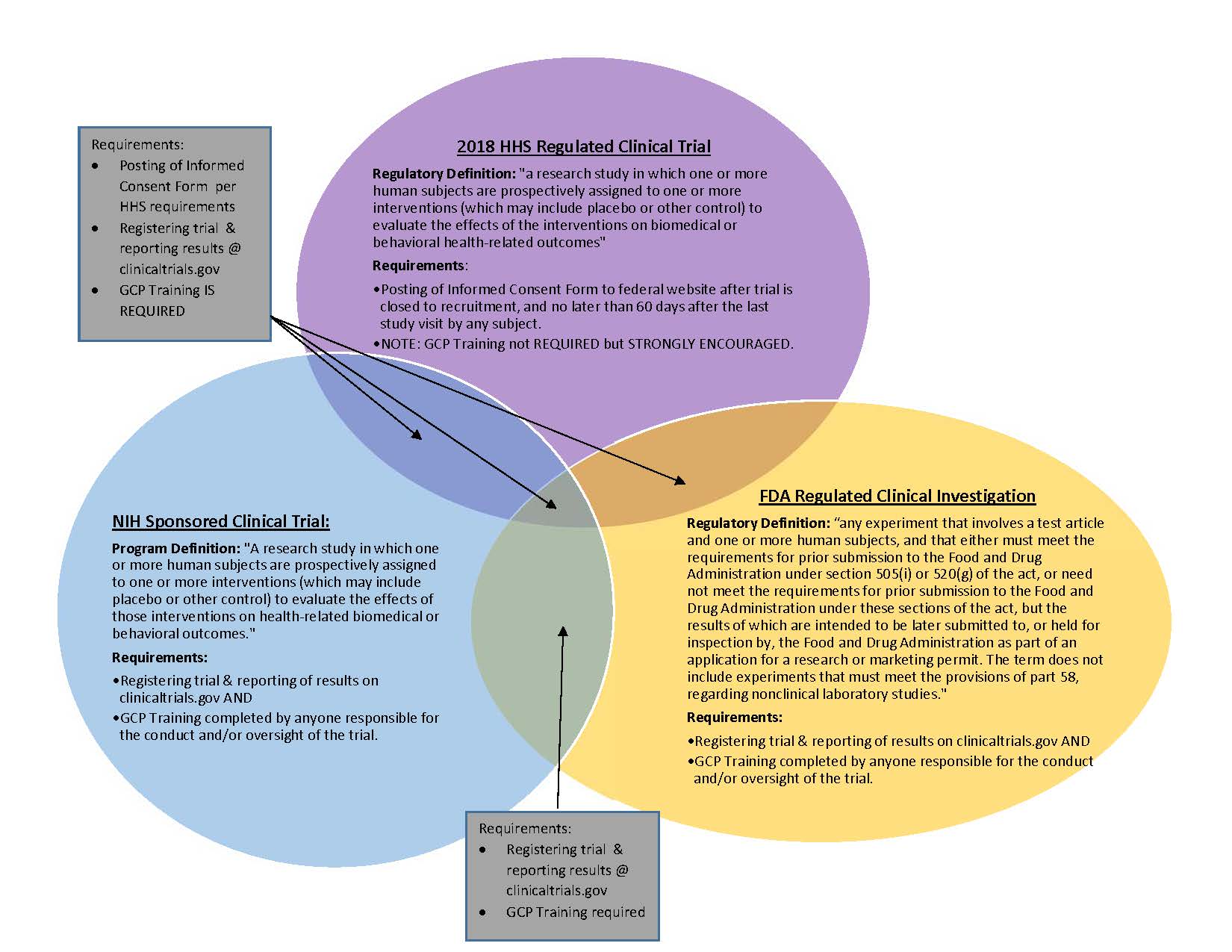
Clinical Trials - Grey Literature in the Health Sciences - Research Guides at University of Alabama - Birmingham

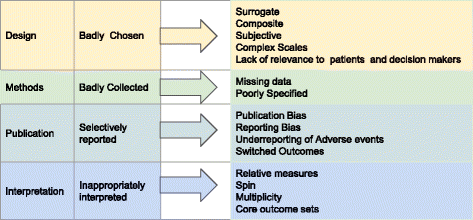
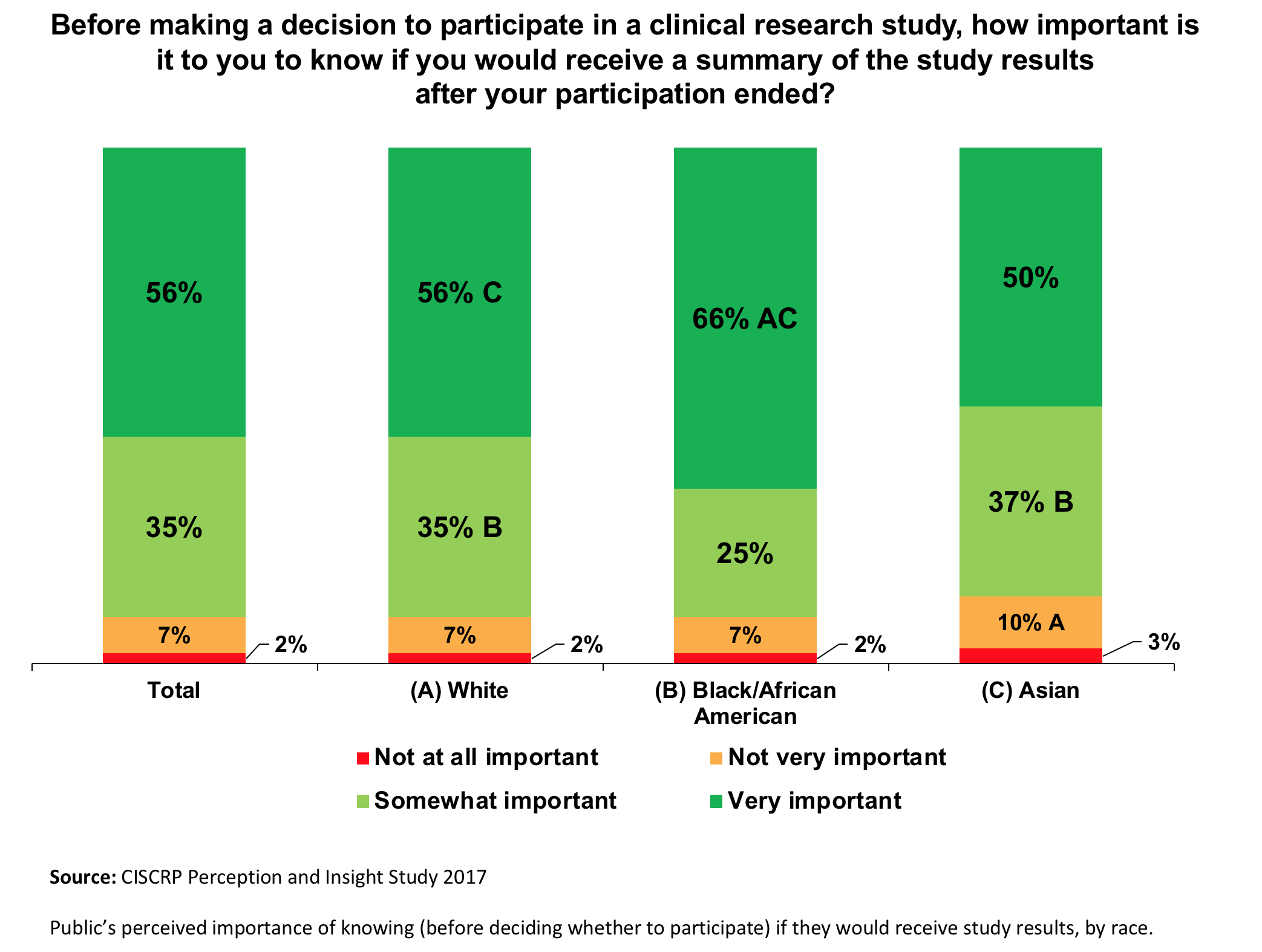
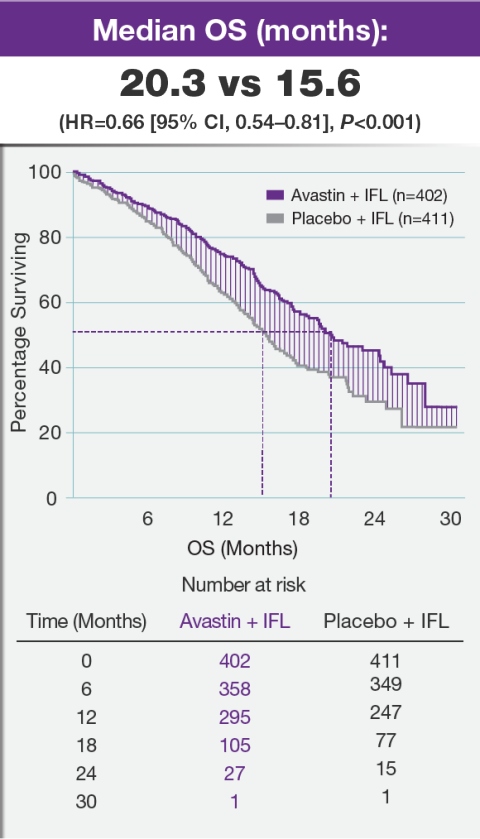
A Guide to Understanding Clinical Trials: Part 2 - Five Factors to Consider When Evaluating Results - Bench Press

The Ontology of Clinical Research (OCRe): An informatics foundation for the science of clinical research - ScienceDirect

Statistical controversies in clinical research: limitations of open-label studies assessing antiangiogenic therapies with regard to evaluation of vascular adverse drug events—a meta-analysis - ScienceDirect
Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data | PLOS Medicine

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet
Evidence for the Selective Reporting of Analyses and Discrepancies in Clinical Trials: A Systematic Review of Cohort Studies of Clinical Trials | PLOS Medicine